Abstract
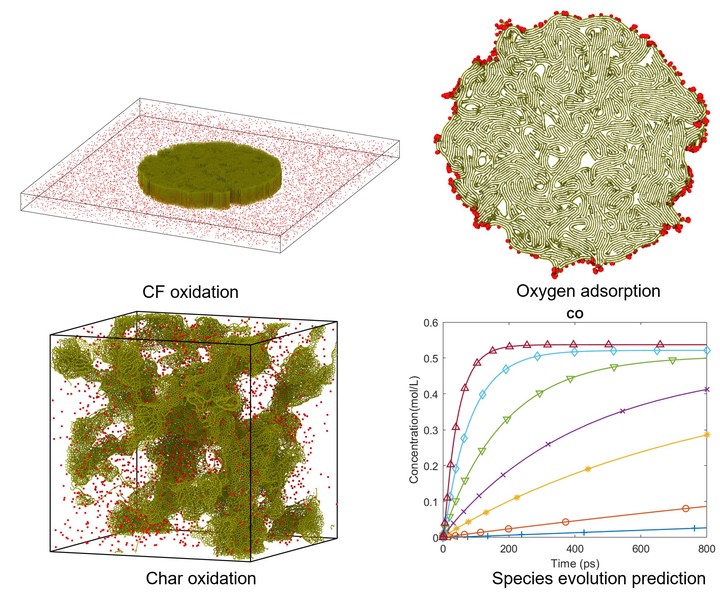
Reactive molecular dynamics (MD) simulations are performed to study the initial stage of the oxidation of carbon fiber and amorphous carbon char with atomic oxygen at temperatures ranging from 1000 K to 4500 K. The carbon fiber and amorphous char models are generated by kinetic Monte-Carlo and liquid quench methods, respectively. The species formed in the simulation are characterized and carbon monoxide is found to be the primary product in both systems. The oxidation results are analyzed in terms of the lifetime of molecules and the reaction rates of various species. Oxygen is found to be adsorbed on the surface of both carbon fiber and amorphous carbon char. Since the amorphous carbon has a significantly larger surface area, the number of oxygen atoms adsorbed on the amorphous carbon surface is significantly higher than on the carbon fiber surface. Six reaction models are proposed to fit the simulation results from which reaction rates at various temperatures are obtained. Reaction rates of the key reactions: carbon oxidation and oxygen adsorption follow the Arrhenius law and the activation energy is extracted for these reactions. These reaction rates are used to predict the long-time evolution of these systems.